Cooperation between researchers in the United States and at the University of Tartu in Estonia helps discover changes in brain tissue that are associated with the very early stages of dementia.
The cause of nearly half of all cases of dementia is Alzheimer’s disease. It is estimated that 47 million people are suffering globally from Alzheimer’s – nearly the population of Spain. Due to the ageing of the population, it is predicted that 131.5 million people will be suffering from Alzheimer’s by 2050 – this is slightly less than the entire population of Russia.
Even though the disease is one of old age, present somewhat more frequently in women than men, it shouldn’t leave anyone indifferent. In addition to deteriorating the patient’s quality of life and the relationships with their loved ones, the disease also has a big impact on the economy. Based on official forecasts, the disease, which is already costing the world billions, will become a trillion-dollar disease in 2018.
The early detection of Alzheimer’s may provide patients the opportunity to live a fuller life and help society save a lot of money. Early detection of the disease, however, is not at all easy. Traditionally, a final diagnosis of Alzheimer’s is only confirmed during an autopsy.
Early detection
Over the past few years, great strides have been made in the treatment and early detection of Alzheimer’s. One important discovery was made in California by Erkki Ruoslahti’s work group, which closely collaborated with Tambet Teesalu’s cancer biology laboratory at the University of Tartu in Estonia.
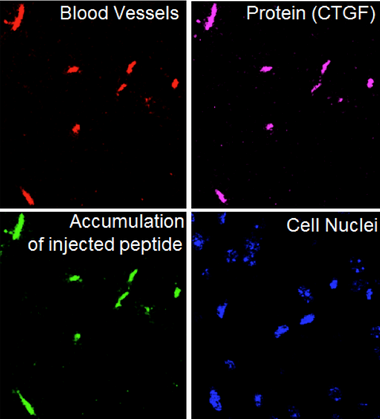 The joint article by Estonian and US researchers, published in the journal Nature Communications, shows that even in the very early stages of Alzheimer’s, a protein called connective tissue growth factor (CTGF) deposits on the blood vessel walls of the brain.
The joint article by Estonian and US researchers, published in the journal Nature Communications, shows that even in the very early stages of Alzheimer’s, a protein called connective tissue growth factor (CTGF) deposits on the blood vessel walls of the brain.
“We discovered that the protein begins to accumulate in blood vessels in the brain even prior to the appearance of amyloid beta plaques,” an Argentinian researcher Pablo Scodeller, one of the lead authors of the study, said. Scodeller, who is also working in Teesalu’s lab, added that if metabolic disorders are already detectable in the brain, it is likely there has been a late start to Alzheimer’s treatment.
Understanding the extent of affected areas
The article describes a short amino acid chain or peptide, which binds to the CTGF protein. After injection into the bloodstream, the peptide DAG seeks out its target protein, CTGF, which, as has already been noted, begins to accumulate in blood vessels of affected areas during the very early stages of Alzheimer’s disease.
This is very important, since the peptide can direct small iron oxide particles (1,000 times smaller than a cell) into the affected area. The accumulation of iron oxide particles can provide contrast in magnetic resonance tomography that helps doctors and researchers understand the extent and localisation of affected areas.
Scodeller said their discovered peptide could detect Alzheimer’s disease much earlier than current clinically approved AD contrast agents. Even though the study in question was performed using mouse models, preliminary testing on clinical tissue samples showed that the peptide binds to Alzheimer’s tissue and CTGF found in humans.

In a time when clinical trials for treating AD continue to fail, researchers are now able to detect an early biomarker of the disease. Scodeller added that the DAG peptide can carry with it not only a diagnostic companion but could also carry a therapeutic drug.
I
This article was originally published in Estonian on the Estonian Public Broadcasting’s science news portal Novaator. It was published in English on Research in Estonia website. This is a lightly edited version. The cover image is illustrative.